LINKED PAPER
Weak asymmetric interspecific aggression and divergent habitat preferences at an elevational contact zone between tropical songbirds. Jones, S.E.I., Tobias, J.A., Freeman, R., Portugal, S.J. 2019. IBIS. DOI: 10.1111/ibi.12793. VIEW
One of the most striking and observable patterns across tropical montane birds are their small elevational distributions. These distributions are typically narrow- often just a few hundred metres in elevation gain- where at their limits they are ‘replaced’ by a similar, but related species. For example, in some of the worlds most species-rich tropical mountains, a hard hike uphill could take you through the entire elevational distributions of several species that appear unable, or at least unwilling, to undertake the same movements themselves. So why do related species live only at specific elevations creating these patterns of elevational zonation?
That ‘replacement’ species are often related, likely holds part of the answer. Related species generally require similar resources, meaning that competition between them can develop at ‘contact zones’ where the upper and lower limits of two species elevational ranges meet. In tropical forest songbirds, such competition is typically expressed in the form of territorial aggression, where a species aggressively defends territorial space with the same vigour towards other species as towards their own species (interspecific aggression). Numerous studies in tropical mountains have found support for interspecific aggression at elevational range edges, and it has even been suggested that competition between species may be the most important factor driving elevational segregation between related species.
 Figure 1 Two species of Catharus Nightingale-Thrush that ‘replace’ one another with changes in elevation: the lower elevation Black-headed Nightingale-Thrush C. mexicanus (left) and the higher elevation Ruddy-capped Nightingale-Thrush C. frantzii (right) © Sam Jones
Figure 1 Two species of Catharus Nightingale-Thrush that ‘replace’ one another with changes in elevation: the lower elevation Black-headed Nightingale-Thrush C. mexicanus (left) and the higher elevation Ruddy-capped Nightingale-Thrush C. frantzii (right) © Sam Jones
Crucially, however, competition between related bird species is usually asymmetric, with one species aggressively dominant over another. This pattern of asymmetry prevails in species that occupy adjacent elevational ranges, in turn presenting an intriguing question- why is it that competitively dominant species do not expand their distributions into the elevational range of another? More specifically, does a preference for other important ecological factors such as microhabitat choice influence this relationship?
We investigated this question in two species of Nightingale-Thrushes that occupy different elevational ranges in the cloud forests of Cusuco National Park, Honduras. To do this, we first mapped territories of the two species (Black-headed Nightingale-Thrushes Catharus mexicanus and Ruddy-capped Nightingale-Thrushes C. frantzii, respectively) to establish their elevational range limits and ‘contact zone’ on the moutainside. After this, we then undertook a series of playback experiments on each territory. These experiments involved subjecting each territorial holding bird to songs of its own species, its ‘replacement’ species (the other species of Nightingale-Thrush), and a control species (to assess whether aggression was not just directed towards any singing bird in the territory). During these experiments, the behavioural responses to each song treatment were recorded (e.g. song output and how quickly birds responded to playback) in order to determine levels of territorial aggression. To determine the microhabitat habitats that each species occupied, we also measured a variety of habitat attributes such a tree types, understory density and soil density in 20 m2 habitat plots across the elevational range limits between the two species.
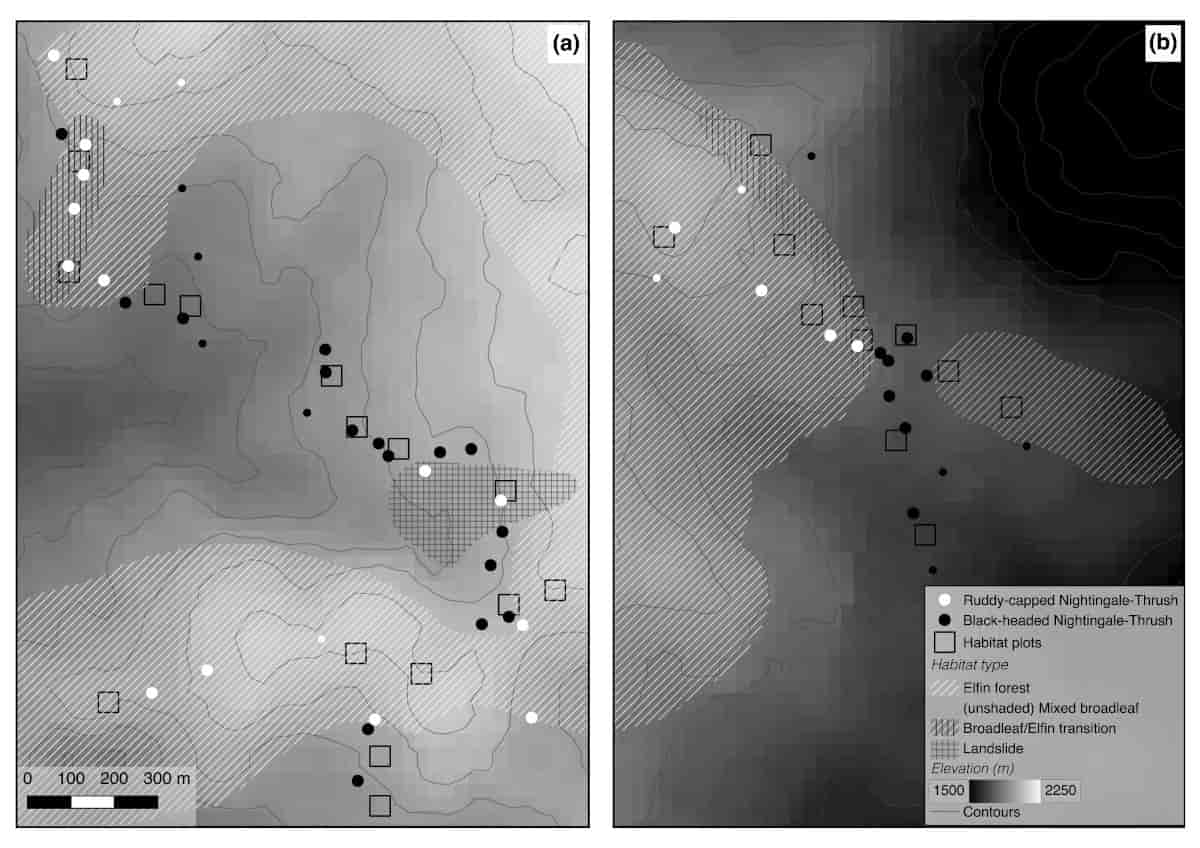 Figure 2 Territory locations in relation to habitat and elevation at two localities in Cusuco National Park, Honduras. Larger points show whether playbacks were conducted on territorial Black-headed Catharus mexicanus or Ruddy-capped Nightingale-Thrushes C. frantzii and smaller points show additional territories of each species at which playback experiments were not conducted. Mapped habitat boundaries away from the immediate vicinity of habitat plots are approximated. Click to view larger
Figure 2 Territory locations in relation to habitat and elevation at two localities in Cusuco National Park, Honduras. Larger points show whether playbacks were conducted on territorial Black-headed Catharus mexicanus or Ruddy-capped Nightingale-Thrushes C. frantzii and smaller points show additional territories of each species at which playback experiments were not conducted. Mapped habitat boundaries away from the immediate vicinity of habitat plots are approximated. Click to view larger
Our study yielded two key findings. Firstly, playback experiments indicated that levels of interspecific aggression differed between the two species, in that Black-headed Nightingale-Thrushes were aggressive towards Ruddy-capped Nightingale-Thrushes, but not vice-versa. This interspecific aggression was restricted to a handful of Black-headed Nightingale-Thrushes in close proximity to the range edge, however. Secondly, in combination with these playback experiments, both species differed in their habitat selection. Territories of Black-headed Nightingale-Thrushes were situated in broadleaf dominated forests, whereas Ruddy-capped Nightingale-Thrushes were found in fern-dominated elfin forests, transitions between broadleaf and elfin forests (with occasional bamboo stands) and succession scrub dominated by pine.
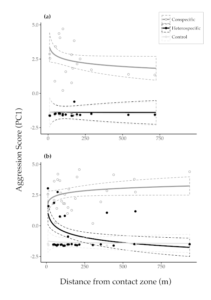
Figure 3 Territorial aggression as estimated as a composite of behavioural responses to playback on territories against distance from the contact zone in (a) Ruddy-capped Nightingale-Thrushes Catharus frantzii and (b) Black-headed Nightingale-Thrushes C. mexicanus. Data points show responses to playback experiments of conspecific, heterospecific and control treatments (higher PC1 scores denote stronger aggression). Solid lines show GLMM model fits (log-transformed); dashed lines show confidence intervals. The overlapping confidence intervals for control and heterospecific trials in Ruddy-capped Nightingale-Thrushes illustrate the lack of interspecific aggression observed in experiments on this species (compare with Black-headed Nightingale-Thrushes)
Taken together, these results suggest that interspecific aggression cannot act alone to explain their elevational range limits and that habitat selection is also likely to be a key process driving their elevational segregation. Nonetheless, our results are consistent with the hypothesis that a dominant species may relegate a subordinate species to less desirable habitat (competitive exclusion). In the case of our study, the lower elevation Black-headed Nightingale-Thrushes appear to resign their higher elevation counterparts (Ruddy-capped Nightingale-Thrushes) into habitats that they do not want to occupy. Exactly what it is about these specific habitats that makes them more appealing to one species, but not another is not immediately obvious and requires further study.
Irrespective of the exact ecological reason behind these habitat choices, understanding the drivers of elevational range restriction has taken grave importance in the quest to understand the consequences of changing climates on sensitive tropical ecosytems. Numerous studies, including at our field site, have displayed upslope shifts in the elevational ranges of tropical species. On the one hand, the onward march of competitively dominant lower elevation species may push their subordinate high elevation replacements into increasingly smaller areas in an ‘escalator to extinction’. On the other hand, however, it may be that habitats themselves offer a safe haven from upslope movements, assuming of course that habitats (or other features of the habitats such as prey sources) do not shift further upslope themselves. Many questions remain to be answered in what drives the patterns of diversity on tropical mountains, but more broadly, our study highlights the importance of examining factors in unison to understand the distributional patterns of species.
Nominate this article for a BOU Science Communication Award.
Further reading
The ‘escalator to extinction’- displaying how upslope shifts in the elevational ranges of species causes some mountain top species to go locally extinct:
Freeman, B.G., Scholer, M.N., Ruiz-Gutierrez, V. & Fitzpatrick, J.W. 2018. Climate change causes upslope shifts and mountaintop extirpations in a tropical bird community. Proceedings of the National Academy of Sciences 115: 11982-11987. VIEW
Dominance patterns between montane species (note the specific relationships displayed between tropical birds)- see therein for references to a number of experimental field studies:
Freeman, B.G. 2019. Lower elevation animal species do not tend to be better competitors than their higher elevation relatives. Global Ecology and Biogeography DOI: 10.1111/geb.13014 VIEW
Upslope shifts in the elevational ranges of birds in Cusuco National Park, Honduras over the past decade:
Neate-Clegg, M.H.C., Jones, S.E.I., Burdekin, O., Jocque, M. & Şekercioğlu, Ç.H. 2018. Elevational changes in the avian community of a Mesoamerican cloud forest park. Biotropica 50: 805–815. VIEW
An example of how competition between related species may also drive habitat preferences and local species range patterns in lowland tropical species:
Robinson, S.K. & Terborgh, J. 1995. Interspecific aggression and habitat selection by Amazonian birds. Journal of Animal Ecology 64: 1–11. VIEW
A background on the exceptional biological diversity of species in mountains globally (and particularly in tropical mountains), and some of the reasons that promote this exceptional diversity:
Rahbek, C., Borregaard, M.K., Colwell, R.K., Dalsgaard, B., Ben G Holt, Morueta-Holme, N., Nogues-Bravo, D., Whittaker, R.J. & Fjeldså, J. 2019. Humboldt’s enigma: What causes global patterns of mountain biodiversity? Science 365: 1108–1113. VIEW
Image credit
Featured image: Black-headed Nightingale-Thrush Catharus mexicanus © Sam Jones




